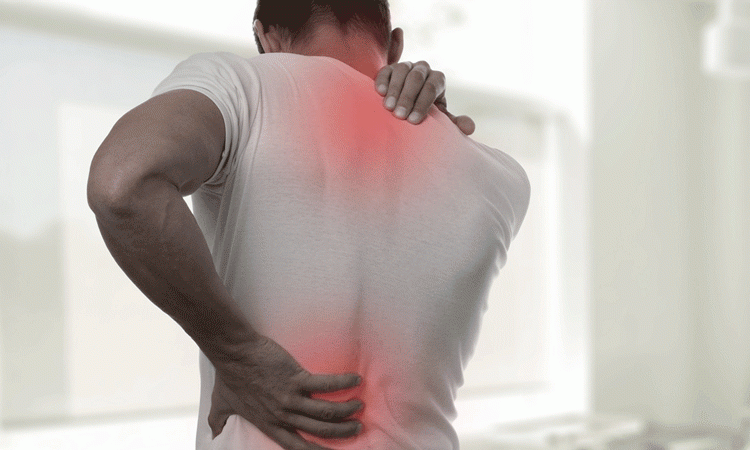
Long-term efficacy and safety of secukinumab in patients with psoriatic arthritis: 5-year (end-of-study) results from the phase 3 FUTURE 2 study - The Lancet Rheumatology
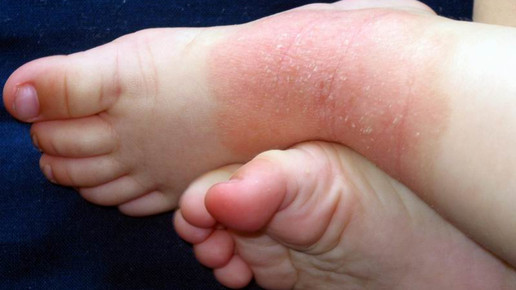
Novartis Cosentyx receives EU approval for first-line systemic treatment in pediatric psoriasis | World Pharma Today
II-079 V013 Gebrauchsinformation: Information für Anwender Cosentyx® 150 mg Injektionslösung in einem Fertigpen Secukinumab L
J&J's Tremfya grabs psoriatic arthritis nod, setting up a battle with Cosentyx, Taltz and more | Fierce Pharma

Cosentyx (Secukinumab): PHASE III-Studie zeigt positive Langzeitergebnisse für AS- und PsA-Patienten
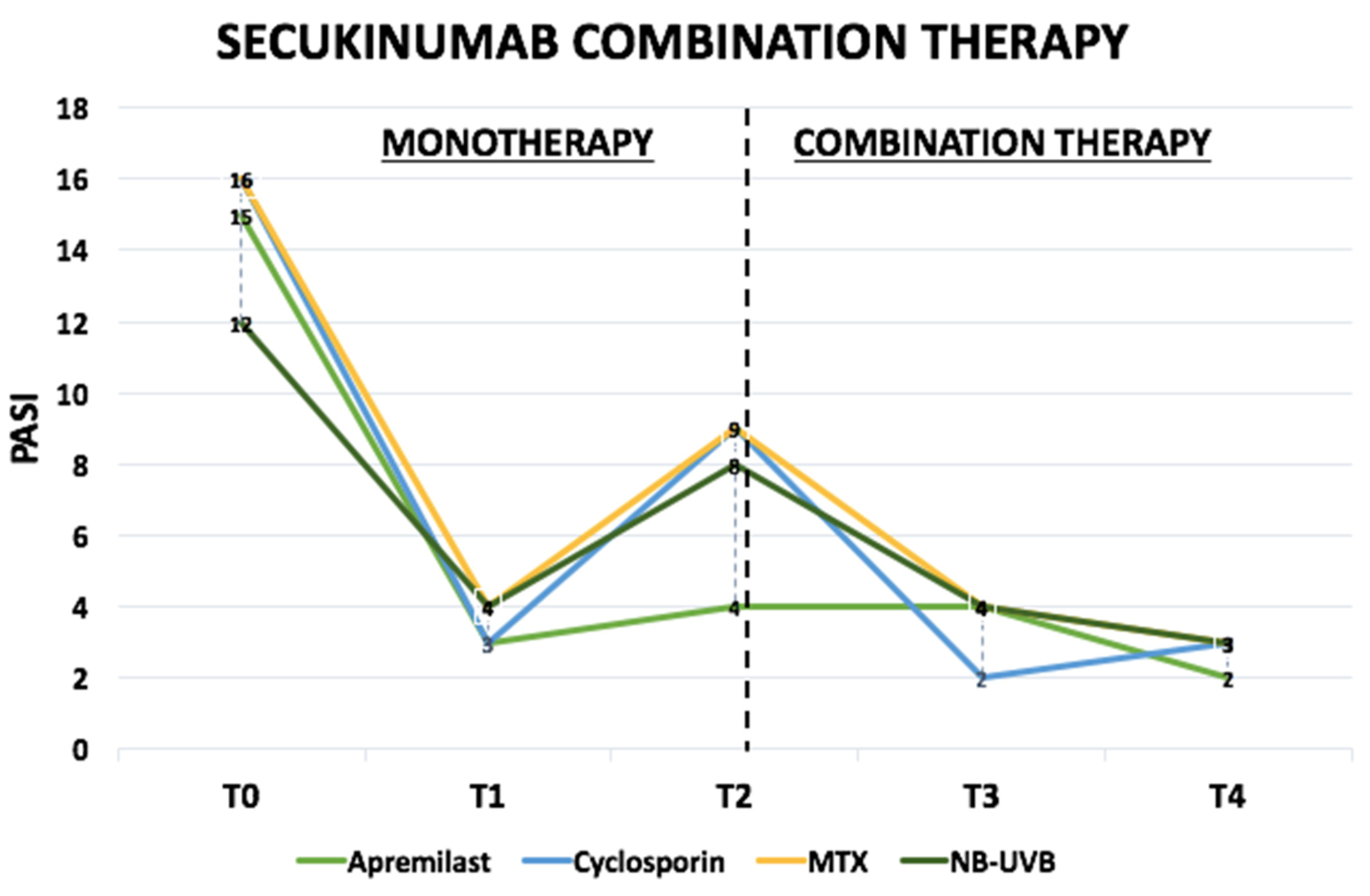
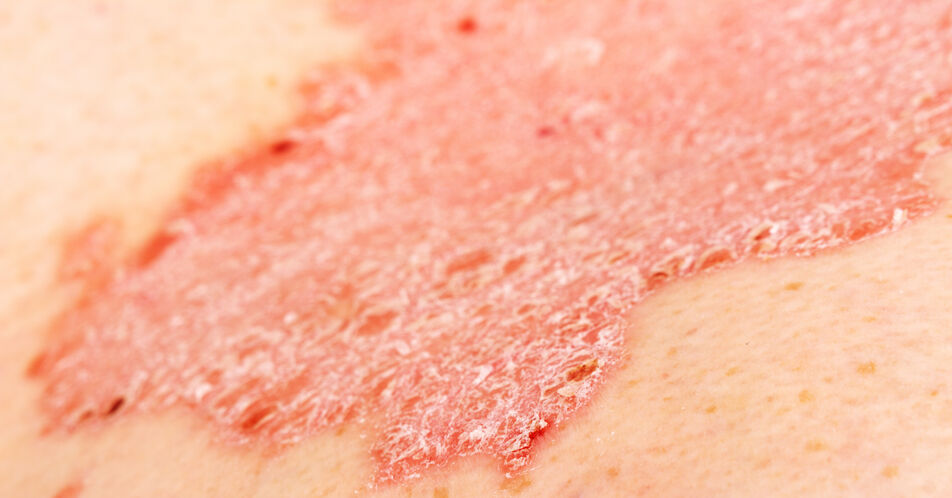
Pharmaceuticals | Free Full-Text | Secukinumab Loss of Efficacy Is Perfectly Counteracted by the Introduction of Combination Therapy (Rescue Therapy): Data from a Multicenter Real-Life Study in a Cohort of Italian Psoriatic